Knowing the differences between techniques for efficient lab work in condenser chemistry is key. This article aims to help you understand two essential processes: distillation and reflux. Although they both use condensers, their purposes and applications in chemical laboratories are distinct – ranging from simple to complex fractional distillation. Let’s explore these differences in the context of condenser chemistry.
 |
 |
Understanding Distillation in Condenser Chemistry
Distillation, a cornerstone in condenser chemistry, is a method used to separate mixtures based on differences in their volatilities in a boiling liquid mixture. This technique, which can be as straightforward as simple distillation or as complex as vacuum distillation, involves heating a liquid until it vaporises and then cooling the vapour, often using a Liebig or Dimroth condenser prior to the launch of effective air condensers, to create a separate liquid phase. This technique is vital in purifying liquids and separating mixtures into their individual components, particularly when dealing with high boiling points.
Components of Distillation
- Boiling Vessel: Contains the mixture to be distilled, often connected with ground glass joints for secure fitting.
- Heating Source: Provides the energy to vaporize the liquid. In methods like steam distillation, this involves passing steam through the mixture.
- Condenser: Types of condenser used include the CondenSyn®, Liebig, Graham, or Allihn condenser, which cools the vapour back into liquid.
- Collection Vessel: Collects the distilled liquid, also known as the condensed liquid.
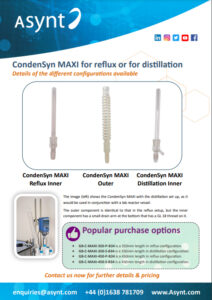
The CondenSyn Distillation Adapter
Applications of Distillation
Distillation, including fractional distillation and rotary evaporation, is critical in pharmaceuticals (where it’s used to purify counter medications), petrochemicals, alcohol production, and more. It’s an indispensable tool in condenser chemistry for ensuring product purity and quality.
Reflux in Condenser Chemistry
Reflux, often used in synthesis and reaction control in condenser chemistry, involves boiling a reaction mixture and condensing the vapour back into the vessel through a reflux condenser. This process allows for the continuous cycling of volatile components without loss, enabling reactions to proceed for extended periods at elevated temperatures, often using a fractionating column or air condenser for more efficient separation.
Asynt – CondenSyn MAXI configuration guide for reflux or distillation setup V1.0
Components of Reflux
- Reaction Vessel: Contains the reactants and solvent, sometimes featuring a flexible tube for pressure adjustments.
- Heating Source: Sustains the boiling of the mixture.
- Condenser: Returns the vapour to the liquid state, cycling it back into the vessel. Graham and Dimroth condensers are common choices where CondenSyn is not available.
Applications of Reflux
Reflux is fundamental in organic synthesis and other chemical reactions where precise temperature control and extended reaction times are necessary. It’s a staple in condenser chemistry, facilitating complex reaction schemes and product formations.
Comparing Distillation and Reflux
While both distillation and reflux are integral to condenser chemistry, they serve different purposes. Distillation, from simple to vacuum distillation, is primarily used for separation and purification, while reflux, often utilising a fractionating column, promotes and controls chemical reactions. The equipment used in both processes may appear similar, notably the condenser, but their roles and the conditions under which they operate differ significantly.
Key Differences
- Purpose: Distillation separates components based on volatility, whereas reflux allows a reaction to proceed under controlled conditions.
- Outcome: The outcome of distillation is a purified product, while reflux leads to reaction completion or product formation.
- Duration: Distillation, whether using a simple or a more complex setup like rotary evaporators, is typically a shorter process than reflux’s often extended duration.
Looking for Condenser Solutions?
Understanding the differences between distillation and reflux in condenser chemistry is essential for anyone engaged in laboratory work. But understanding is only half the equation. You also need the right equipment for the job.
At Asynt, we offer high-quality condensers and related apparatus designed to carry out distillation and reflux processes with precision and efficiency. In order to advance these practices in a more sustainable manner, our CondenSyn® waterless air condenser replaces traditional water condensers, addressing environmental concerns and reducing common lab risks. It features a unique hyperbolic profile made from borosilicate glass, which ensures optimal heat removal. Offering clear visibility and easy maintenance, these condensers demonstrate our innovation in condenser chemistry, catering to various laboratory needs.
“Our CondenSyn® waterless condensers are rapidly becoming a necessity in laboratories all over the world, driven by their ease of use and massive associated water savings benefits compared to using traditional water condensers. Not only has the CondenSyn proved itself as a sustainable alternative for reflux reactions but increasingly also for distillation experiments using the CondenSyn Distillation Adapter. A typical Chemistry laboratory replacing their standard water condensers with CondenSyn units can expect to see a return on its investment in as little as six months.“
– Dr. Ffion McKeague, Operations Director at Asynt.
We invite you to explore our catalogue and discover the ideal condenser chemistry solutions for your laboratory needs, from small benchtop scales to those suited to work with larger lab reactors. We’re here to support you in your work to advance the frontiers of chemical research and industry applications with the right tools and knowledge.