
You may have seen the acronym “CSTR” in relation to our fReactor modules, or in Flow Chemistry articles, but what are they and, more importantly, how does a CSTR work and why should you care? This blog aims to answer these questions and more!
Click here to skip through to further reading and published papers
What does the CSTR acronym stand for?
CSTR stands for Continuous Stirred Tank Reactor. CSTR and standard batch reactor are two types of chemical reactors that are commonly used in industry and research.
What are the benefits of using a CSTR over a standard batch reactor?
Here are some of the basic benefits of using a CSTR over a standard batch reactor:
- Continuous operation: A CSTR operates continuously, which means that the reaction mixture is constantly flowing through the reactor. This allows for more efficient use of resources and can result in higher yields.
- Consistent product quality: Because a CSTR operates continuously, it can maintain a more consistent product quality compared to a batch reactor, which can have variations in product quality from batch to batch.
- Better heat transfer: CSTRs have better heat transfer properties than batch reactors, which means that heat can be more easily and efficiently removed from the reaction mixture. This can help to prevent overheating and improve reaction efficiency.
- More precise control: A CSTR can be more precisely controlled than a batch reactor, which can result in more accurate and reproducible results. For example, the flow rate of reactants and catalysts can be adjusted to optimize the reaction conditions.
- Scalability: CSTRs can be easily scaled up or down depending on the production requirements, making them a flexible choice for industrial applications.
How does a CSTR actually work?
Reactant concentrations can be easily monitored and adjusted by simply controlling the input/output flow rates into/out of the reactor, which gives chemists greater accuracy when carrying out complex reactions. The stirring of solutions within the reactor also ensures that reactants remain evenly mixed, resulting in consistent products across different batches.
Furthermore, since reactions occur in real time with CSTRs, their residence times are much shorter compared to batch processing methods. This shortens reaction times significantly and results in increased throughputs which help speed up overall production processes and make them more cost-effective for manufacturers.
The fReactor from the University of Leeds and Asynt, uses a specific type of CSTR that has been designed to be compact and easy to use. It features a modular design that allows users to quickly and easily change the reactor size and configuration to suit their specific needs. Additionally, the fReactor can be used with a range of different types of stirrers and heating systems, making it a versatile choice for a wide range of applications.
This short video, below, demonstrates clearly how effective the mixing is with the fReactor CSTR modules:
Understanding the physics of cascade CSTRs and the fReactor Classic
Overall, continuous stirred tank reactors provide great benefits to chemists by allowing for more precise control over reactant concentrations, more efficient stirring for even distribution, and shortened reaction times compared to traditional batch-processing methods. With their improved capabilities over conventional systems, it is easy to see why CSTRs have become so popular among today’s chemists.
To understand how the fReactor Classic utilises cascade CSTRs we would draw your attention to the dedicated website administered by the inventors of the platform at the iPRD, University of Leeds; Prof. Nikil Kapur and Prof. John Blacker. Here they offer a wealth of information and resources, including the fReactor Modeller pictured below.
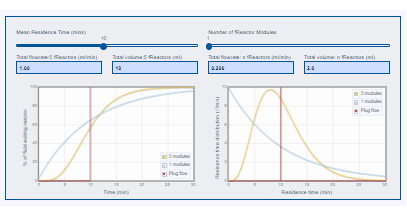 Click the image to navigate to fReactor.com and the interactive modeller above
Click the image to navigate to fReactor.com and the interactive modeller above
The team explain how the cascade of these CSTRs in fReactor works to provide incredibly effective and accurate mixing:
A single well mixed reactor (a continuous stirred tank reactor – CSTR) means an incoming ‘element’ of fluid is instantaneously mixed into the reaction mixture within the reactor. Since there is a steady flow, then for every incoming ‘element’ there has to be an outgoing ‘element’. With good mixing, important for uniform reaction conditions, there is chance that the incoming ‘element’ almost immediately leaves in the outgoing stream. Equally, the ‘element’ may spend a long time in the reactor. It is just like a bag of balls where you drop a ball in, shake it and immediately select one at random to be the one that leaves. Some balls spend only a small amount of time in there whilst some will spending a long time – it really is just a game of chance and probability.
But by cascading the reactors together minimises the odds of our ‘element’ passing straight through the cascade of reactors. There are well established models that predict this (and the fReactors have been shown to fit this model in this paper ) – the app mentioned above lets you explore these models for yourself.
To find out more about the range of Flow Chemistry options available from Asynt, you can click HERE or contact our experts to discuss any questions or requirements you have.
Further reading:
What is “Green” Flow Chemistry?
 How much of Flow Chemistry in an engineering task?
How much of Flow Chemistry in an engineering task?






